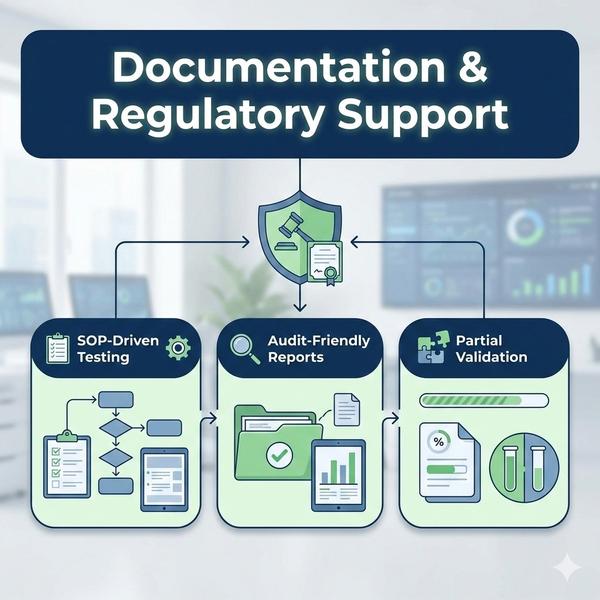
At Dextrose Technologies Pvt. Ltd., we provide Documentation & Regulatory Support to deliver inspection-ready analytical outputs for R&D and QC programs. Our focus is SOP-driven execution, clean data presentation, and structured reporting that speeds up internal QA review and client submissions. We support chromatography and mass spectrometry workflows (HPLC/RP-HPLC, GC-FID, HS-GCMS, GCMS, LCMS—scope dependent) with consistent documentation and clear interpretation. SOP-Driven Testing Testing is performed using controlled workflows with documented sample handling, instrument conditions, and batch execution notes for traceability. Regulatory-Style Analytical Reports We provide structured reports with sample identifiers, method conditions, chromatograms/spectra (as applicable), results tables, calculation basis, and clear conclusions—formatted for review and submission support. Partial Method Validation Support We support partial validation for defined scope parameters such as accuracy, precision, and linearity, with summary tables and acceptance criteria where agreed. Data Review & Interpretation We review chromatograms/spectra to highlight key findings, trends, and anomalies, and provide practical interpretation notes aligned to your decision goal. Client-Specific Reporting Formats If you have internal templates or required sections, we can align reporting to your preferred format for smoother approvals and submissions. Outcome you can expect * Audit-friendly documentation with clear traceability * Faster client submissions supported by structured reporting Get started Share your method, analytes, sample count, and preferred report template. We’ll propose the best documentation package for your workflow.